Multiple Choice
Identify the
letter of the choice that best completes the statement or answers the question.
|
|
| 1. | Which
term most precisely describes the cellular process of breaking down large molecules into smaller
ones? a. | catalysis | b. | metabolism | c. | anabolism | d. | dehydration | e. | catabolism | | |
|
|
| 2. | Which
of the following is (are) true for anabolic pathways? a. | They do not
depend on enzymes. | b. | They are highly regulated sequences of chemical
reactions. | c. | They consume energy to build up polymers from
monomers. | d. | They release energy as they degrade polymers to
monomers. | e. | both B and C | | |
|
|
| 3. | Which
of the following is a statement of the first law of thermodynamics? a. | Energy cannot be
created or destroyed. | b. | The entropy of the universe is
decreasing. | c. | The entropy of the universe is
constant. | d. | Kinetic energy is stored energy that results from the specific
arrangement of matter. | e. | Energy cannot be transferred or
transformed. | | |
|
|
| 4. | The
first law of thermodynamics states that energy can be neither created nor destroyed. For living
organisms, which of the following is an important consequence of the first law? a. | The energy
content of an organism is constant. | b. | The organism ultimately must obtain all of the necessary energy
for life from its environment. | c. | The entropy of an organism decreases with time as the organism
grows in complexity. | d. | Organisms are unable to transform
energy. | e. | Life does not obey the first law of
thermodynamics. | | |
|
|
| 5. | Living organisms increase in complexity as they grow, resulting in a decrease in the
entropy of an organism. How does this relate to the second law of thermodynamics? a. | Living organisms
do not obey the second law of thermodynamics, which states that entropy must increase with
time. | b. | Life obeys the second law of thermodynamics because the
decrease in entropy as the organism grows is balanced by an increase in the entropy of the
universe. | c. | Living organisms do not follow the laws of
thermodynamics. | d. | As a consequence of growing, organisms create more disorder in
their environment than the decrease in entropy associated with their
growth. | e. | Living organisms are able to transform energy into
entropy. | | |
|
|
| 6. | Which
of the following statements is a logical consequence of the second law of
thermodynamics? a. | If the entropy
of a system increases, there must be a corresponding decrease in the entropy of the
universe. | b. | If there is an increase in the energy of a system, there must
be a corresponding decrease in the energy of the rest of the universe. | c. | Every energy
transfer requires activation energy from the environment. | d. | Every chemical
reaction must increase the total entropy of the universe. | e. | Energy can be
transferred or transformed, but it cannot be created or destroyed. | | |
|
|
| 7. | Which
of the following types of reactions would decrease the entropy within a cell? a. | dehydration
reactions | b. | hydrolysis | c. | respiration | d. | digestion | e. | catabolism | | |
|
|
| 8. | According to the second law of thermodynamics, which of the following statements is
incorrect? a. | The synthesis of
large molecules from small molecules is exergonic. | b. | Earth is an open
system. | c. | Life exists at the expense of energy derived from its
environment. | d. | A living cell can never function as a closed
system. | e. | Every chemical reaction in a cell results in a loss of free
energy. | | |
|
|
| 9. | The
mathematical expression for the change in free energy of a system is:  G = G =  H - T H - T S. Which of the following is (are)
incorrect? S. Which of the following is (are)
incorrect? a. |  S is the change in entropy, a measure
of randomness. S is the change in entropy, a measure
of randomness. | b. |  H is the change in enthalpy, the energy available to do
work. H is the change in enthalpy, the energy available to do
work. | c. |  G is the change in free energy. G is the change in free energy. | d. | T is the
absolute temperature. | e. | both A and B | | |
|
|
| 10. | What
is the change in free energy of a system at chemical equilibrium? a. | slightly
increasing | b. | greatly increasing | c. | slightly
decreasing | d. | greatly decreasing | e. | no net
change | | |
|
|
| 11. | Which
of the following is true for all exergonic reactions? a. | The products
have more total energy than the reactants. | b. | The reaction proceeds with a net release of free
energy. | c. | Some reactants will be converted to
products. | d. | A net input of energy from the surroundings is required for the
reactions to proceed. | e. | The reactions are nonspontaneous. | | |
|
|
| 12. | When
glucose monomers are joined together by glycosidic linkages to form a cellulose polymer, the changes
in free energy, total energy, and entropy are as follows:
|
|
| 13. | A
chemical reaction that has a positive  G is correctly described as G is correctly described as a. | endergonic. | b. | endothermic. | c. | enthalpic. | d. | spontaneous. | e. | exothermic. | | |
|
|
| 14. | Why
is ATP an important molecule in metabolism? a. | Its hydrolysis provides an input of free energy for exergonic
reactions. | b. | It provides energy coupling between exergonic and endergonic
reactions. | c. | Its terminal phosphate group contains a strong covalent bond
that when hydrolyzed releases free energy. | d. | A and B only | e. | A, B and
C | | |
|
|
| 15. | The
hydrolysis of ATP to ADP and inorganic phosphate (ATP + H2O  ADP + Pi ) ADP + Pi ) a. | has a  G of about -7 kcal/mol under standard
conditions. G of about -7 kcal/mol under standard
conditions. | b. | involves hydrolysis of a terminal phosphate bond of
ATP. | c. | can occur
spontaneously under appropriate conditions. | d. | Only A and B are
correct. | e. | A, B, and C are correct. | | |
|
|
| 16. | ATP
generally energizes a cellular process by a. | releasing heat upon hydrolysis. | b. | acting as a
catalyst. | c. | coupling free energy released by ATP hydrolysis to free energy
needed by other reactions. | d. | breaking a high-energy bond. | e. | binding directly
to the substrate(s) of the enzyme. | | |
|
|
| 17. | Which
of the following is most similar in structure to ATP? a. | an anabolic
steroid | b. | a DNA helix | c. | RNA
nucleotides | d. | an amino acid with three phosphate groups
attached | e. | a phospholipid | | |
|
|
| 18. | Which
of the following statements is true concerning catabolic pathways? a. | They combine
molecules into more energy-rich molecules. | b. | They are usually coupled with anabolic pathways to which they
supply energy in the form of ATP. | c. | They are endergonic. | d. | They are
spontaneous and do not need enzyme catalysis. | e. | They build up
complex molecules such as protein from simpler compounds. | | |
|
|
| 19. | Which
of the following statements is (are) true about enzyme-catalyzed reactions? a. | The reaction is
faster than the same reaction in the absence of the enzyme. | b. | The free energy
change of the reaction is the same as the reaction in the absence of the
enzyme. | c. | The reaction always goes in the direction toward chemical
equilibrium. | d. | A and B only | e. | A, B, and
C | | |
|
|
| 20. | How
can one increase the rate of a chemical reaction? a. | Increase the activation energy
needed. | b. | Cool the reactants. | c. | Decrease the
concentration of the reactants. | d. | Add a catalyst. | e. | Increase the
entropy of the reactants. | | |
|
|
| 21. | Sucrose is a disaccharide, composed of the monosaccharides glucose and fructose. The
hydrolysis of sucrose by the enzyme sucrase results in a. | bringing glucose
and fructose together to form sucrose. | b. | the release of water from sucrose as the bond between glucose
and fructose is broken. | c. | breaking the bond between glucose and fructose and forming new
bonds from the atoms of water. | d. | production of water from the sugar as bonds are broken between
the glucose monomers. | e. | utilization of water as a covalent bond is formed between
glucose and fructose to form sucrase. | | |
|
|
| 22. | Reactants capable of interacting to form products in a chemical reaction must first
overcome a thermodynamic barrier known as the reaction's a. | entropy. | b. | activation energy. | c. | endothermic
level. | d. | heat content. | e. | free-energy
content. | | |
|
|
| 23. | Which
of the following statements regarding enzymes is true? a. | Enzymes decrease
the free energy change of a reaction. | b. | Enzymes increase the rate of a
reaction. | c. | Enzymes change the direction of chemical
reactions. | d. | Enzymes are permanently altered by the reactions they
catalyze. | e. | Enzymes prevent changes in substrate
concentrations. | | |
|
|
| 24. | Which
of the following is not true of enzymes? a. | Enzyme catalysis is dependent on the pH and temperature of the
reaction environment. | b. | Enzyme catalysis is dependent on the three-dimensional
structure or conformation of the enzyme. | c. | Enzymes provide activation energy for the reaction they
catalyze. | d. | Enzymes are composed primarily of protein, but they may bind
nonprotein cofactors. | e. | Enzyme activity can be inhibited if the enzyme's allosteric
site is bound with a noncompetitive inhibitor. | | |
|
|
| 25. | During a laboratory experiment, you discover that an enzyme-catalyzed reaction has a
 G of -20 kcal/mol. If you double the
amount of enzyme in the reaction, what will be the G of -20 kcal/mol. If you double the
amount of enzyme in the reaction, what will be the  G for the new reaction? G for the new reaction? a. | -40 kcal/mol | b. | -20
kcal/mol | c. | 0 kcal/mol | d. | +20
kcal/mol | e. | +40 kcal/mol | | |
|
|
| 26. | The
active site of an enzyme is the region that a. | binds allosteric regulators of the
enzyme. | b. | is involved in the catalytic reaction of the
enzyme. | c. | binds the products of the catalytic
reaction. | d. | is inhibited by the presence of a coenzyme or a
cofactor. | e. | both A and B | | |
|
|
| 27. | According to the induced fit hypothesis of enzyme catalysis, which of the following is
CORRECT? a. | The binding of
the substrate depends on the shape of the active site. | b. | Some enzymes
change their structure when activators bind to the enzyme. | c. | A competitive
inhibitor can outcompete the substrate for the active site. | d. | The binding of
the substrate changes the shape of the enzyme's active site. | e. | The active site
creates a microenvironment ideal for the reaction. | | |
|
|
|
Refer to the figure below to answer the following
questions.
|
|
| 28. | Which
curve represents the behavior of an enzyme taken from a bacterium that lives in hot springs at
temperatures of 70°C or higher? a. | curve 1 | b. | curve
2 | c. | curve
3 | d. | curve
4 | e. | curve
5 | | |
|
|
| 29. | Which
curve was most likely generated from analysis of an enzyme from a human stomach where conditions are
strongly acid? a. | curve
1 | b. | curve
2 | c. | curve
3 | d. | curve
4 | e. | curve
5 | | |
|
|
| 30. | What
is a nonprotein "helper" of an enzyme molecule called? a. | accessory
enzyme | b. | allosteric group | c. | coenzyme | d. | functional group | e. | enzyme
activator | | |
|
|
| 31. | Zinc,
an essential trace element for most organisms, is present in the active site of the enzyme
carboxypeptidase. The zinc most likely functions as a(n) a. | competitive
inhibitor of the enzyme. | b. | noncompetitive inhibitor of the
enzyme. | c. | allosteric activator of the enzyme. | d. | cofactor
necessary for enzyme activity. | e. | coenzyme derived from a vitamin. | | |
|
|
| 32. | Consider the following: Succinate dehydrogenase catalyzes the conversion of succinate
to fumarate. The reaction is inhibited by malonic acid, which resembles succinate but cannot be acted
upon by succinate dehydrogenase. Increasing the ratio of succinate to malonic acid reduces the
inhibitory effect of malonic acid. Which of the following is correct? a. | Succinate
dehydrogenase is the enzyme, and fumarate is the substrate. | b. | Succinate
dehydrogenase is the enzyme, and malonic acid is the substrate. | c. | Succinate is the
substrate, and fumarate is the product. | d. | Fumarate is the product, and malonic acid is a noncompetitive
inhibitor. | e. | Malonic acid is the product, and fumarate is a competitive
inhibitor. | | |
|
|
|
The following questions are based on the reaction A + B 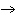 C + D shown in the figure
below. C + D shown in the figure
below.
|
|
| 33. | Which
of the following terms best describes the reaction? a. | endergonic | b. | exergonic | c. | anabolic | d. | allosteric | e. | nonspontaneous | | |
|
|
| 34. | Which
of the following represents the  G of the reaction? G of the reaction?
|
|
| 35. | Which
of the following bests describes the reaction? a. | negative  G, spontaneous G, spontaneous | b. | positive  G, nonspontaneous G, nonspontaneous | c. | positive  G, exergonic G, exergonic | d. | negative  G, endergonic G, endergonic | e. |  G of zero, chemical equilibrium G of zero, chemical equilibrium | | |
|
|
| 36. | Which
of the following represents the activation energy required for the enzyme-catalyzed
reaction?
|
|
| 37. | Which
of the following represents the activation energy required for a noncatalyzed
reaction?
|
|
| 38. | The
mechanism in which the end product of a metabolic pathway inhibits an earlier step in the pathway is
known as a. | metabolic
inhibition. | b. | feedback inhibition. | c. | allosteric
inhibition. | d. | noncooperative inhibition. | e. | reversible
inhibition. | | |
|
|
| 39. | How
does a non-competitive inhibitor decrease the rate of an enzyme reaction? a. | by binding at
the active site of the enzyme | b. | by changing the structure of the
enzyme | c. | by changing the free energy change of the
reaction | d. | by acting as a coenzyme for the
reaction | e. | by decreasing the activation energy of the
reaction | | |
|
|
|
Use the figure below to answer the following
questions.
|
|
| 40. | Which
of the following statements correctly indicate(s) the role of ATP or ADP as an allosteric regulator?
Assume that the supply of energy for cellular processes is adjusted to meet cellular demand for
energy. a. | ATP is an
allosteric inhibitor of catabolic pathways. | b. | ADP is an
allosteric activator of catabolic pathways. | c. | ATP is an
allosteric activator of anabolic pathways. | d. | A and B only | e. | A, B, and
C | | |
|